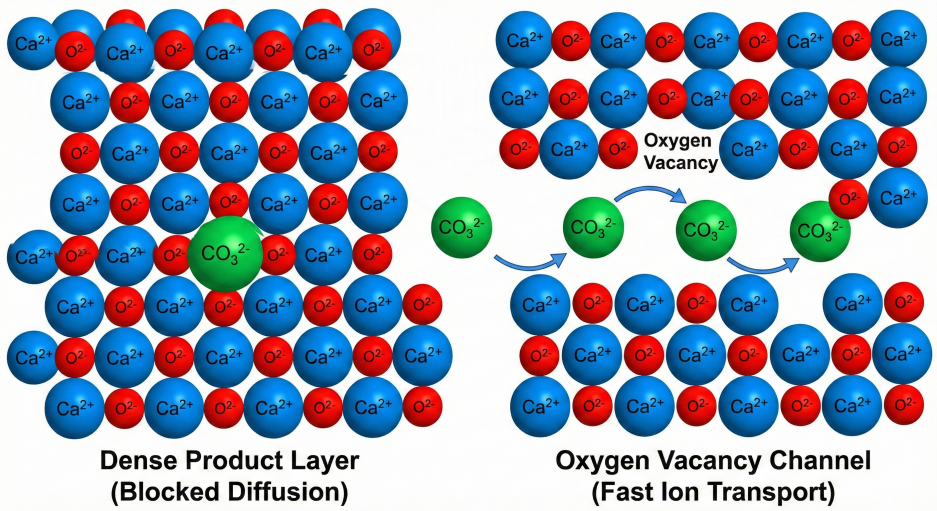
Calcium looping (CaL) is a great way to store thermal energy because it holds a lot of energy. But there is a big problem: CaO adsorbents quickly lose their activity. When they get hot, they sinter. Also, the product layer blocks diffusion. Because of these issues, we cannot easily use CaL on a large scale right now. To fix this, researchers are adding oxygen vacancies at the atomic level. This paper reviews how oxygen vacancies change CaO-based materials for the better. First, we look at how people make these vacancies in the lab. For example, they use aliovalent doping or change the material's shape. We also list the main tools used to check them. Next, the paper explains the science behind this using Density Functional Theory (DFT) calculations. These calculations show us exactly how the vacancies work. They give ions an easier path to travel, which speeds up the physical movement (better kinetics). At the same time, the vacancies act as Lewis basic sites. They grab CO₂ molecules tighter, which helps the reaction happen (better thermodynamics). Finally, we talk about the remaining hurdles, especially keeping the materials stable at very high heat. We suggest that future work should combine live testing (in-situ) with data-driven computer models to build much stronger energy storage materials.